Studies on clinical pharmacy practice, ADR monitoring, and patient safety
Research on drug regulations, ethics, and policy frameworks
Support for regulatory documentation & industry compliance
Pharmacovigilance and real-world evidence studies for better therapeutic outcomes
Patient counseling models and community-based healthcare research
Training students in Good Clinical Practice (GCP) and ethical research standards

At Chennupati Indo-American School of Pharmacy, our research areas represent the core of our academic vision and innovation strategy. We focus on a diverse spectrum of disciplines, including pharmaceutics, pharmacology, biotechnology, regulatory science, and quality control, to drive scientific excellence and real-world impact. Through these research areas, students and faculty collaborate to address critical healthcare challenges, from designing novel drug delivery systems to improving drug efficacy and safety.
Our research ecosystem nurtures interdisciplinary collaboration—bringing together chemists, pharmacologists, biotechnologists, and formulation scientists to work on groundbreaking projects. We emphasize hands-on experimentation, data-driven learning, and ethical inquiry, enabling our researchers to translate ideas into practical solutions that benefit public health. In addition, we actively promote student-led research, providing mentorship, laboratory access, and funding opportunities so that emerging scientists can contribute meaningfully from early in their academic journey.
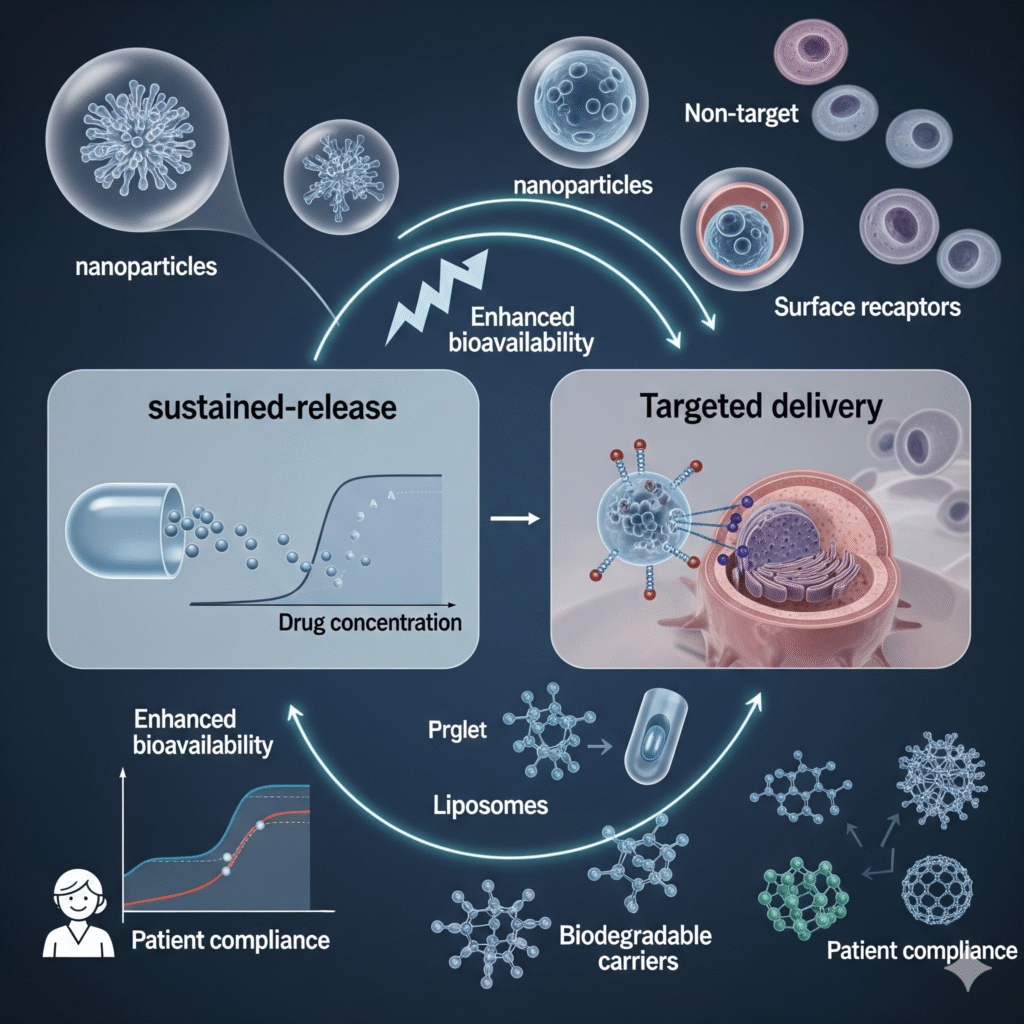
Novel Drug Delivery Systems (NDDS)
-
Sustained & Targeted Delivery
Development of dosage systems for prolonged action and site-specific drug release.
-
Nanotechnology in Formulations
Research on nanoparticles, liposomes, and biodegradable carriers.
-
Improved Bioavailability
Enhancing drug absorption, patient compliance, and therapeutic outcomes.
-
Advanced Drug Routes
Mucoadhesive, transdermal, and alternative delivery methods for better absorption.
-
Stability & In-Vitro Studies
Lorem Ipsum is simply dummy text of the printing and typesetting industry. Lorem Ipsum has been the industry's standard dummy text ever since the 1500s.
Pharmacological Screening & Toxicology
Our research emphasizes understanding drug action, efficacy, and safety through systematic screening and toxicological evaluation.
Focus Areas:
Drug Action & Safety Studies – Experimental models for evaluating pharmacological responses.
Preclinical Evaluation – Use of animal simulators and ethical alternatives for predictive research.
Toxicology Studies – Assessment of herbal and synthetic compounds for safety.
Activity Screening – Investigating neuropharmacological, cardiovascular, and anti-inflammatory potential.
Dose–Response & Mechanism Studies – Validating therapeutic efficacy and mode of action.
Safety & Compliance – Ensuring adherence to ethical guidelines and regulatory standards.

Clinical & Regulatory Research

Enabling Infrastructure

-
Advanced Analytical Labs
Equipped with HPLC, UV-Vis spectrophotometers, dissolution apparatus, and viscometers for precision drug analysis.
-
Animal Simulator Lab
Ethical training and research in pharmacology & toxicology without animal use.
-
Herbal Drug Lab
Focused on phytochemical standardization, extraction, and herbal formulation studies.
-
R&D Incubation Center (IASP-Catalyst)
Supports student startups, innovation projects, and patent filing.
-
Library & E-Resources
Access to global journals, e-books, databases, and digital learning platforms.
Subscribe & Follow us
Subscribe & Get More Information
Receive the latest news, event invitations, and academic insights from our school—straight to your inbox. Follow us online to stay connected.
Academics
Science & Tech
Student Resources
Quick Links
Copyright © 2025, Chennupati Indo-American School of Pharmacy. All rights reserved.

