Welcome to the Research Division of Chennupati Indo-American School of Pharmacy (CIASP), a key contributor to Pharmaceutical Research & NDDS India. Our mission is to advance global drug delivery systems and pharmaceutical sciences through pioneering research, industry collaborations, and rigorous analysis, ensuring that we translate fundamental scientific discoveries into practical, innovative solutions that significantly improve patient outcomes globally. Our dedicated research strategy is highly collaborative, involving partnerships with leading national and international universities, pharmaceutical companies, and government funding agencies to ensure our projects remain at the cutting edge of innovation and maintain direct relevance to current industry and societal health needs.
Novel Drug Delivery Systems (NDDS)
Our dedicated research in NDDS focuses on overcoming the limitations of conventional therapies. We are actively exploring biodegradable polymers and advanced nanotechnology to enhance drug solubility, stability, and permeability, making us a key center for specialized NDDS Research in India. These efforts aim not just to improve bioavailability but also to minimize patient toxicity and dosage frequency, advancing patient-centered drug delivery
Phytochemistry & Herbal Drug Formulation
Through meticulous isolation and characterization using advanced equipment like HPLC and GC-MS, we ensure high standards for herbal extracts. Our focus is on the sustainable use of medicinal plants and the integration of traditional knowledge with modern science to develop safe, standardized herbal formulations with enhanced therapeutic value.
Pharmaceutical Analysis & Method Validation
Compliance is paramount. We adhere strictly to international pharmacopeial standards (USP, BP, IP) to guarantee quality benchmarks. Our comprehensive method validation covers accuracy, precision, specificity, and robustness, ensuring that all analytical results are reliable, reproducible, and fully compliant with regulatory bodies like the FDA and ICH.
Industrial Pharmacy & Formulation Technology
Beyond laboratory work, we emphasize industrial relevance. Our research includes critical scale-up and process optimization studies, ensuring that successful lab formulations can be smoothly translated into large-scale commercial production. This training, combined with strict adherence to GMP, QC, and QA systems, prepares our students and researchers for diverse roles in the pharmaceutical industry.
Novel Drug Delivery Systems (NDDS)
NDDS provide controlled, targeted, and efficient drug delivery compared to conventional methods, improving bioavailability, reducing side effects, and enhancing patient compliance.
Research Focus at Our Institution
Targeted Delivery: Nanoparticles, liposomes, microspheres.
Controlled/Sustained Release: Oral, transdermal, injectable systems.
Biodegradable Polymers: Eco-friendly, biocompatible carriers.
Nanotechnology: Enhancing solubility, stability, permeability.
Personalized Delivery: Patient-specific formulations.
Impact & Goals
Minimize toxicity & dosage frequency.
Improve therapeutic outcomes.
Advance global patient-centered drug delivery.
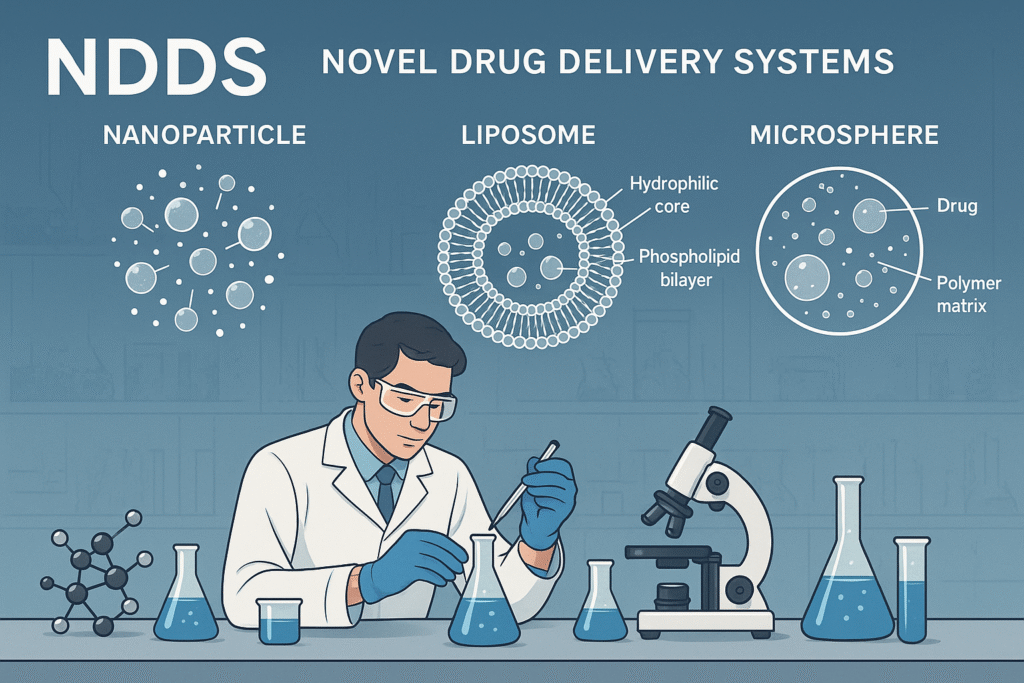
Objectives:
Isolation & Characterization: Phytochemicals studied via HPLC, GC-MS, NMR.
Standardization: Quality control for extracts and formulations.
Formulation Development: Herbal dosage forms—tablets, syrups, ointments.
Pharmacological Evaluation: Anti-inflammatory, antioxidant, antimicrobial, anticancer studies.
Sustainability: Conservation and sustainable use of medicinal plants.
Key Areas:
Plant-based drug discovery
Standardized herbal formulations
Integration of traditional and modern medicine
Enhancing bioavailability & stability

-
Formulation & Process Development
Covers preformulation studies, formulation of tablets, capsules, injections, ointments, and optimization of processes like granulation, compression, and coating.
-
Manufacturing & Quality Systems
Industrial-scale production, packaging, scale-up, and strict compliance with GMP, Quality Control (QC), and Quality Assurance (QA).
-
Advanced Drug Delivery Technologies
Research and development of innovative dosage systems including nanoparticles, liposomes, and sustained-release formulation
-
Career & Professional Opportunities
Diverse roles such as Formulation Scientist, Production Manager, QC Analyst, Regulatory Affairs Officer, and R&D Scientist.

Subscribe & Follow us
Subscribe & Get More Information
Receive the latest news, event invitations, and academic insights from our school—straight to your inbox. Follow us online to stay connected.
Academics
Science & Tech
Student Resources
Quick Links
Copyright © 2025, Chennupati Indo-American School of Pharmacy. All rights reserved.


